| MrMortonScience |
3.2.7 Metal Extraction
Energetics - Kinetics - Equilibria - Redox - Group VII - Group II - Metal Extraction - Haloalkanes - Alkenes - Alcohols - Analytical Tech... - Unit 1
3.2.7 Metal Extraction
Economic factors and recycling
The occurrence of metals in the Earth's crust
· Metals are found in the Earth's crust. The least reactive metals are found as elements (i.e. native, e.g. silver, gold), but most metals are found in compounds, usually oxides or sulphides.
· Rocks that contain a high enough percentage of a metal to be extracted commercially are known as ores (what percentage is required depends on the value of the metal).
· Aluminium and iron are the most abundant metals in the Earth's crust, and they are widely distributed around the world, which explains why they are so widely used.
· There are some other commonly used metals, such as Ni and Cu that are scarce in the Earth's crust, but occur in high grade ores in a few locations.
Methods of extraction
· Most compounds are found as oxides or sulphides, but the sulphides are usually converted to oxides before extraction. The extraction of metals involves reduction, usually of metal oxides. This reduction of the metal compound is usually done in one of three main ways:
Reduction of metal oxides with carbon
Nearly all metals can be extracted this way, but many are not because:
Extraction of iron in the Blast furnace
Economic factors and recycling
The occurrence of metals in the Earth's crust
· Metals are found in the Earth's crust. The least reactive metals are found as elements (i.e. native, e.g. silver, gold), but most metals are found in compounds, usually oxides or sulphides.
· Rocks that contain a high enough percentage of a metal to be extracted commercially are known as ores (what percentage is required depends on the value of the metal).
· Aluminium and iron are the most abundant metals in the Earth's crust, and they are widely distributed around the world, which explains why they are so widely used.
· There are some other commonly used metals, such as Ni and Cu that are scarce in the Earth's crust, but occur in high grade ores in a few locations.
Methods of extraction
· Most compounds are found as oxides or sulphides, but the sulphides are usually converted to oxides before extraction. The extraction of metals involves reduction, usually of metal oxides. This reduction of the metal compound is usually done in one of three main ways:
- by heating with carbon (in the form of coke)
- by heating with a more reactive metal (active metal)
- by electrolysis
- ·the energy requirements (electricity and / or heat)
- the cost of the reductant (carbon is cheap but other reactants may not be)
- the metal purity required (the higher the required purity, the greater the cost in obtaining that purity)
Reduction of metal oxides with carbon
Nearly all metals can be extracted this way, but many are not because:
- Metal carbides can be formed (e.g. Titanium/TiC, Tungsten/WC) giving metals undesirable properties
- The temperature required is excessively high for reactive metals
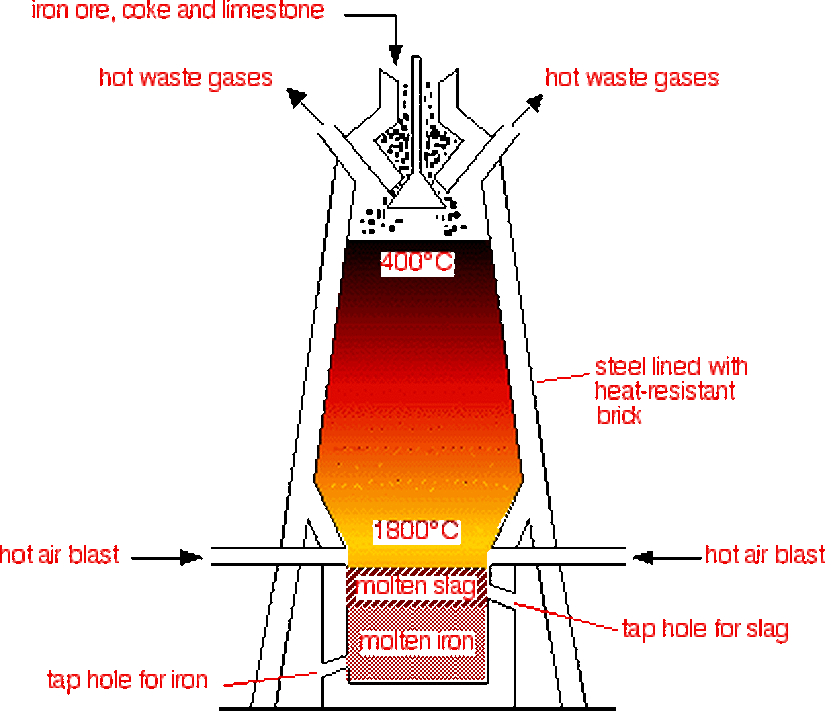
Extraction of iron in the Blast furnace
- Heamatite is the main ore of iron and contains Fe2O3.
- This is a continuous process.
- Raw materials: iron ore, coke (a form of carbon), hot air and limestone.
Steps
- In the high temperatures in the furnace, more coke is reacted with the carbon dioxide produced, forming CO.
- The iron oxide is reduced to iron by the CO and some C (due to the heat the iron is molten, and so can be run off).
- The limestone (calcium carbonate) decomposes (splits up into simpler substances) in the heat.
- The calcium oxide (a base) reacts with acidic impurities such as sand (silicon dioxide), producing a molten mixture of compounds, mainly calcium silicate, called slag which floats on the iron – slag is used in construction (e.g. road making, cement making).
Formulae
- C(s) + O2(g) → CO2(g)
- C(s) + CO2(g) → 2 CO(g)
- Fe2O3(s) + 3 CO(g) → 2 Fe(l) + 3 CO2(g)
- Fe2O3(s) + 3 C(s) → 2 Fe(l) + 3 CO(g)
- CaCO3(s) → CaO(s) + CO2(g)
- CaO(s) + SiO2(s) → CaSiO3(s)
Problems
- Pollution problems arise from using carbon, giving CO2 (a greenhouse gas) and CO (toxic)
- Roasting of sulphide ores to give oxides leads to SO2 pollution, leading to acid rain.
Making Steel
The iron produced (cast or pig iron) has impurities such as C (making it very brittle), P and S.
These impurities are removed in the Basic Oxygen Process, which is a batch process.
1) S is removed by injecting powdered Mg - this forms MgS which floats on the surface and can be scraped off (S is not removed by the oxygen as the iron would be oxidised instead).
2) Scrap steel is added (to recycle it and to lower the temperature in the converter).
3) Oxygen is blasted through the mixture, turning impurities to oxides (e.g. Carbon dioxide – lost as gas)
4) Limestone (decomposes to CaO) / CaO is added – reacts with solid acidic oxides – forming molten slag
5) Other metals or C are added to give steel the desired properties
The iron produced (cast or pig iron) has impurities such as C (making it very brittle), P and S.
These impurities are removed in the Basic Oxygen Process, which is a batch process.
1) S is removed by injecting powdered Mg - this forms MgS which floats on the surface and can be scraped off (S is not removed by the oxygen as the iron would be oxidised instead).
2) Scrap steel is added (to recycle it and to lower the temperature in the converter).
3) Oxygen is blasted through the mixture, turning impurities to oxides (e.g. Carbon dioxide – lost as gas)
4) Limestone (decomposes to CaO) / CaO is added – reacts with solid acidic oxides – forming molten slag
5) Other metals or C are added to give steel the desired properties
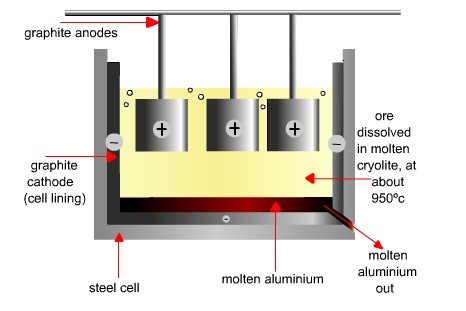
Reduction of metal oxides by electrolysis of melts
- For metals where carbon reduction is not suitable, electrolysis is usually used, e.g. aluminium (electrolysis is not used if the metal has to be very pure).
- The raw materials for the extraction of aluminium are purified aluminium oxide (Al2O3) and cryolite (Na3AlF6).
- The aluminium oxide must be molten or dissolved to conduct electricity, and so is dissolved in molten cryolite (this requires a lower temperature than using molten aluminium oxide).
- The electrodes are made of graphite.
- The positive electrode burns due to the oxygen produced there (forming carbon dioxide), and so the positive electrode has to be replaced frequently.
- The main cost in this process is the electricity – so a cheap supply is needed – plants are often built near hydroelectric power stations.
Reduction of metal halides with metal
- Titanium cannot be extracted with carbon because titanium carbide (TiC) is formed rather than titanium (similar reactions take place for vanadium, tungsten and molybdenum).
- Titanium is a very useful metal as it abundant, has a low density and is corrosion resistant – it is used for making strong, light alloys for use in aircraft for example.
- Titantium has be very pure to have these useful properties and so electrolysis is not suitable. The metal is extracted by reaction with a more reactive metal (e.g. Mg, Na).
- The main ore is rutile, containing TiO2.
Steps
- TiO2 (solid) is converted to TiCl4 (liquid) at 900°C: TiO2 + 2 Cl2 + 2 C → TiCl4 + 2 CO
- The TiCl4 is purified by fractional distillation in an Ar atmosphere.
- The Ti is extracted by Mg or Na in an Ar atmopshere at 500°C: TiCl4 + 4 Na → Ti + 4 NaCl
- TiO2 is converted to TiCl4 as it can be purified by fractional distillation, TiCl4 being molecular (liquid at room temperature) rather than ionic like TiO2 (solid at room temperature).
- The process is expensive due to the heat, the Na/Mg, the Ar, and the need to remove moisture (as TiCl4 is susceptible to hydrolysis).
Economic factors and recycling
Why metals are recycled
Recycling Al and Fe
Problems
Why metals are recycled
- Saves resources (e.g. metal ores)
- Less waste (e.g. mining waste)
- Saves energy resources (less energy to re-cycle than make from ore)
- Reduces air pollution (e.g. CO2 – greenhouse effect, SO2 – acid rain, CO - toxic)
Recycling Al and Fe
- Al is simply melted down.
- Scrap steel is added to iron from the Blast Furnace in the Basic Oxygen Process
Problems
- Separating the metals.
- Need ways to stop people dumping metal.
Practice Exam questions
|
| ||||||||||||
Energetics - Kinetics - Equilibria - Redox - Group VII - Group II - Metal Extraction - Haloalkanes - Alkenes - Alcohols - Analytical Tech... - Unit 1